FDA is Recommending Pausing the Use of Johnson and Johnson Vaccine
FDA is Recommending Pausing the Use of Johnson and Johnson Vaccine in the U.S.
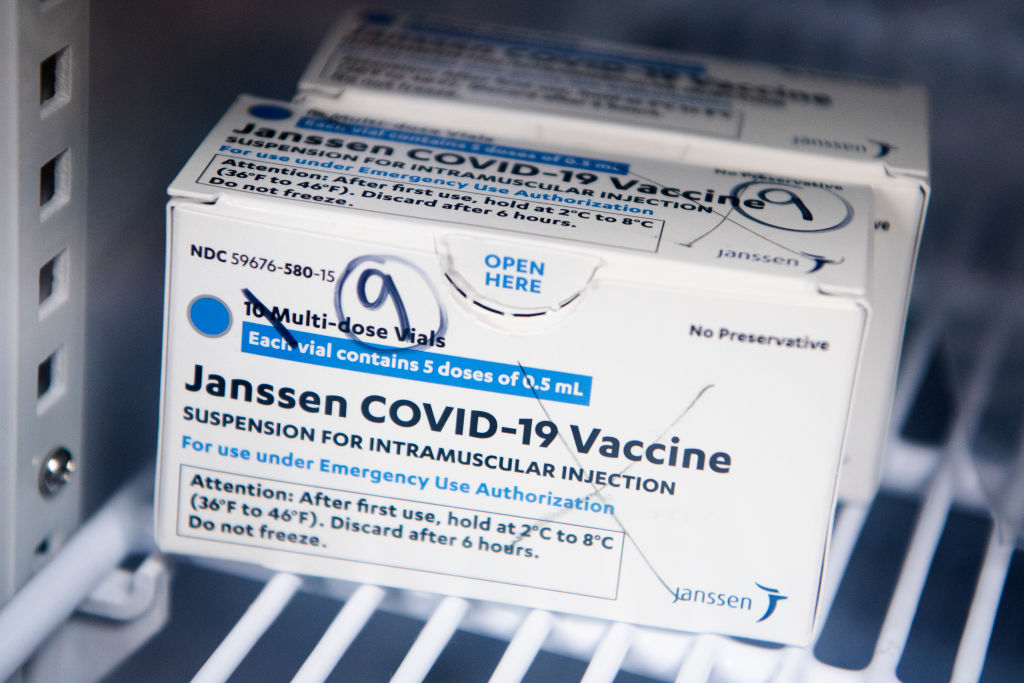
Source: Tom Williams / Getty
The FDA has announced that the distribution of the Johnson & Johnson COVID-19 will be paused after six women in the U.S. had rare blood-clotting after receiving the vaccine. The women were ranging in ages from 18 to 48, and while six people may not seem like a lot precautions are being taken swiftly. There have been approximately 6.8 million shots given with the majority of people not having issues.
The pausing of the Johnson & Johnson vaccine does not affect the other two vaccines from Pfizer and Moderna. But if you did receive the Johnson & Johnson shot and are having to severe stomach pain, leg cramps, or severe headaches you should call your healthcare provider right away. If you do not know what shot you received, check your CDC vaccination card where it is noted what brand shot you received. If you have an appointment scheduled to receive the Johnson & Johnson COVID-19 vaccine, it is advised to call your provider for further directions.
Get Breaking News & Exclusive Contest in Your Inbox:
[ione_media_gallery id=”3917798″ src=”https://newsone.com/” overlay=”true”]
The Latest:
- Trump Says All Presidents Should Be Forced To Take Cognitive Tests
- Speaker Mike Johnson Is Having A Rough Go Of It Amid GOP Collapse
- James Broadnax Executed After Cousin Confesses To Being The Shooter
- Rev. Stephen Green On Faith, Justice, And The Liberating Gospel Of Jesus
- NYC Finally Addressing ‘Deed Theft’ After Ignoring Black Homeowners
- 10 Songs You Might Not Have Known DJ Kid Capri Produced
- Cincinnati Bearcats Unveil Air Jordan 14 PE
- These Twin Sisters Just Graduated No. 1 & No. 2 In Their Class And The Internet Loves Them
- Thousands Expected To Attend May Day Protests Across US
- Defense Secretary Pete Hegseth Grilled By Congress As Iran War Drags On
FDA is Recommending Pausing the Use of Johnson and Johnson Vaccine in the U.S. was originally published on mycolumbusmagic.com













